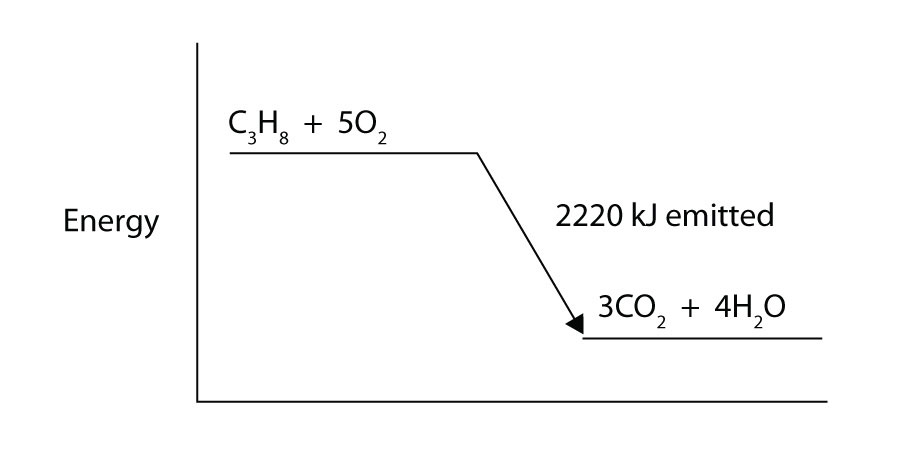
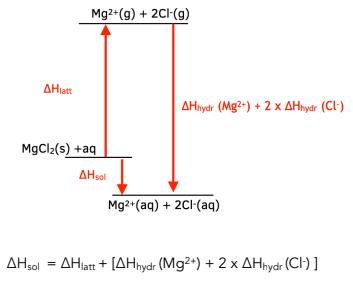
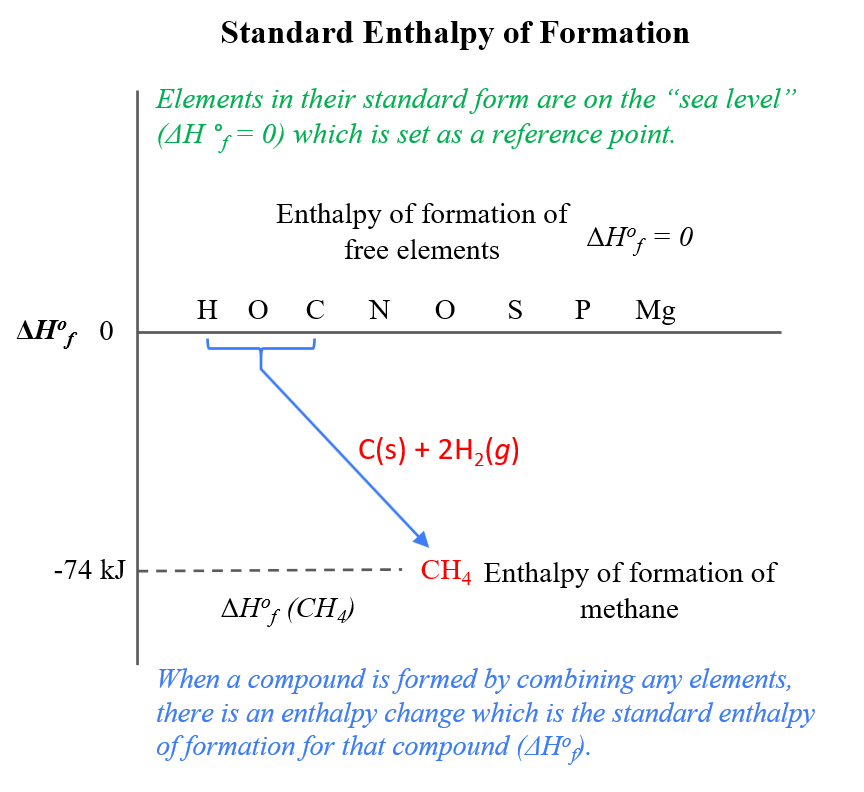
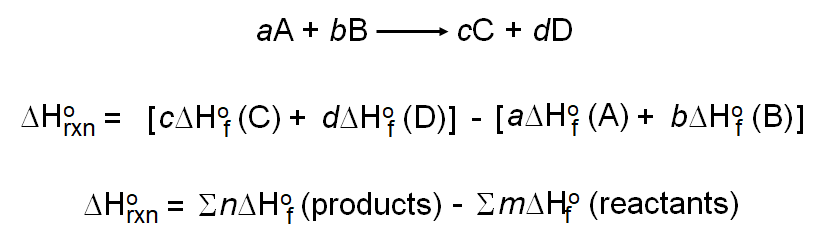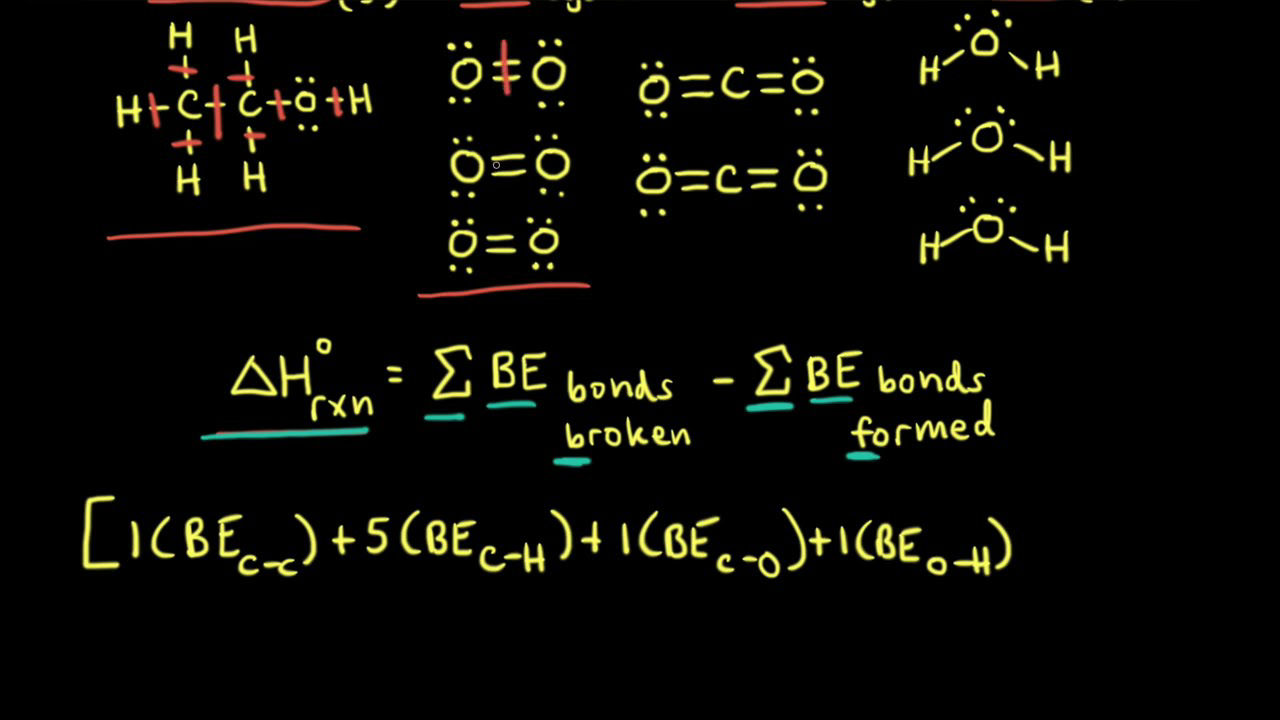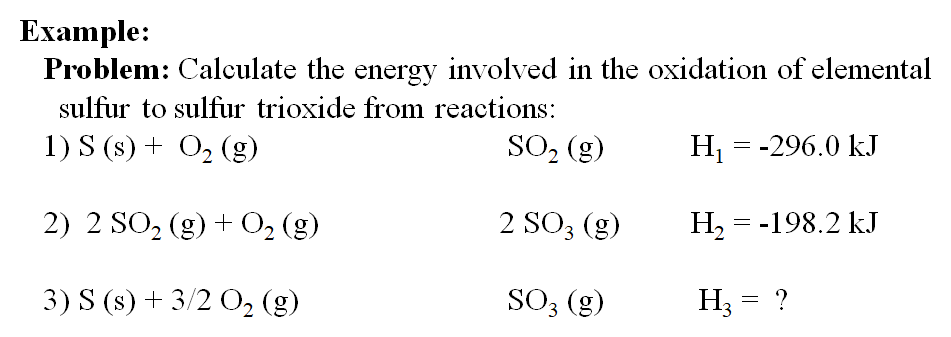Calculate the Standard Enthalpy of the Reaction,From the Following δH° Values - Chemistry | Shaalaa.com

SOLVED: Question 3 Calculate the Enthalpy of the Photosynthesis Reaction 0/1 points Balance the reaction shown below that occurs during photosynthesis and use bond energies to estimate the enthalpy change for the